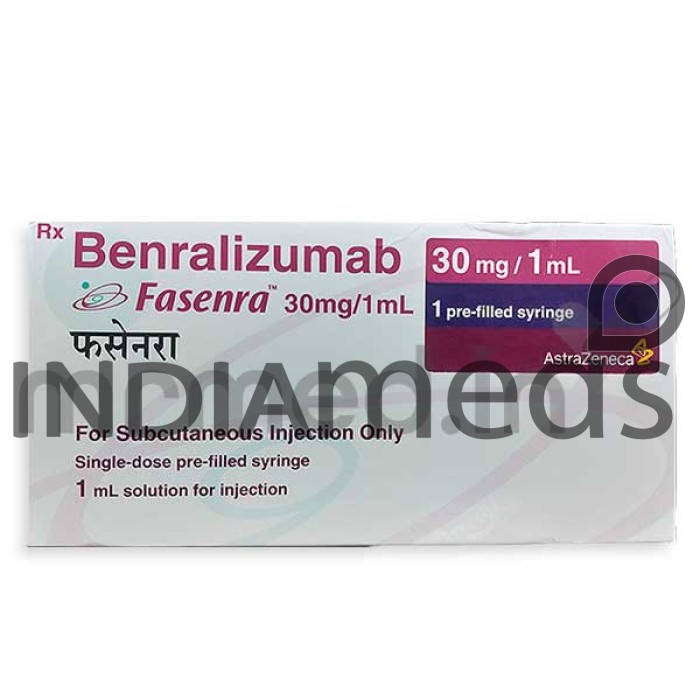
Fasenra 30mg Injection 1ml: Effective Treatment for Your Condition
₹265 883
Нашли данный товар дешевле?
Характеристики:
(смотреть все)
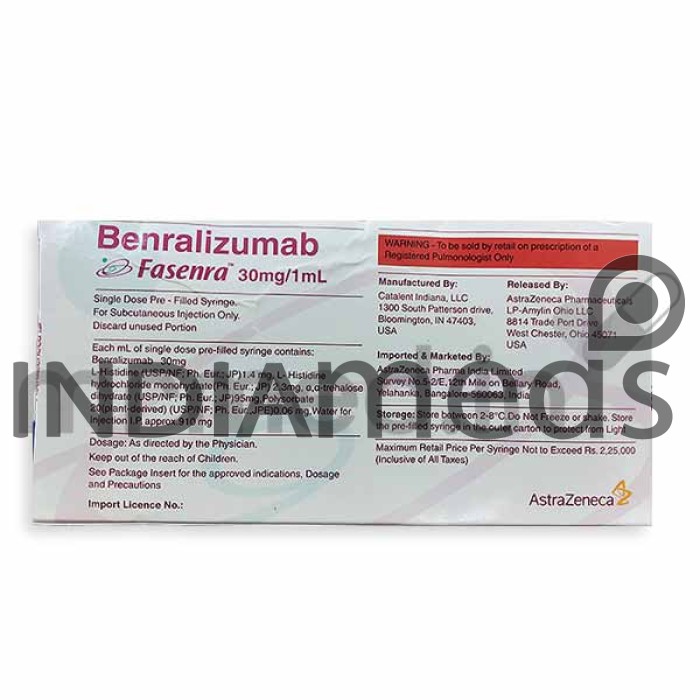
Manufacturer
ASTRAZENECA PHARMA INDIA LTD
Origin of Medicine
United Kingdom
Price for
1 Prefilled Syringe(s) Of 1ml
Salt Composition
Benralizumab
Delivery
Delivery from India within 15 days.
Payment
Pay quickly and conveniently
Cumulative discounts
Collect bonuses and gifts








-200x200.jpg)